SODIUM-HF: Sodium restriction in patients with heart failure
By Hans Haag with editorial support from Ricky Turgeon
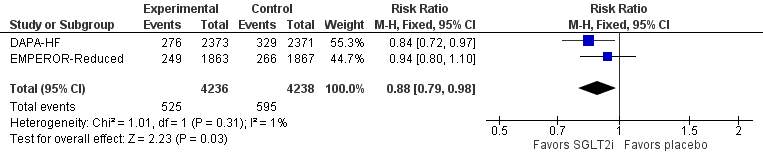
SODIUM-HF. Lancet 2022;399:1391-400.
Bottom line: In patients with heart failure (HF), achieved dietary sodium restriction ~1600 mg/day did not reduce the risk of death or cardiovascular hospitalizations/ED visits compared with usual care achieving sodium about 2000 mg/day.
Patients (n=841 randomized, 806 analyzed)
Included:
Adults with clinical HF
NYHA class 2-3
Receiving optimal guideline-directed medical therapy
Key exclusions:
Dietary sodium intake <1500 mg/day
Serum sodium <130 mmol/L
eGFR <20 mL/min/1.73 m^2 or requiring hemodialysis
Within 1 month of cardiovascular hospitalization
Baseline
Age 67, 33% female, 58% Canadian
NYHA 2 (71.4%), NYHA 3 (26.9%), LVEF median 36%, NT-proBNP ~800, HF hospitalization in last 12 months ~33%
Comorbidities: HTN 62%, CAD 47%, AF/AFL 39%, diabetes 33%
SBP 118, eGFR 61
Meds: ACEI/ARB/ARNI 81%, beta-blocker 87%, MRA 57%
Diuretic use not reported
Baseline median sodium intake ~2200 mg/day (self-report)
Intervention: Dietary sodium intake <1500 mg/d
Country-specific meal plans & menus
Achieved ~1600 mg/d at 12 months (approximately 400 mg/d lower than comparator group)
Comparator: Usual care
General advice to restrict dietary sodium (as provided during routine clinical practice)
Achieved ~2000 mg/d at 12 months
Outcomes @ 12 months
No difference for any of the clinical outcomes
Improvements in some subjective secondary outcomes with the intervention
QoL: Mean difference in Kansas City Cardiomyopathy Questionnaire +3.4/100 vs placebo
NYHA class 1: 16% vs 11% (odds ratio 1.69 [95% CI 1.16-2.50] for improvement ≥1 NYHA class)
6min walk distance (6MWD): +6.6 meters (95% CI -9 to +22)
Internal validity
Low risk of bias for clinical outcomes (death, hospitalizations)
Low risk of selection bias: Independent statistician generated randomization list; allocation concealment by automated web-based system. Block randomization according to the study site.
Low risk of performance & detection bias: Patients and clinicians unblinded to treatment allocation, but relatively objective outcomes & blinded endpoint adjudication
Attrition bias: ITT analysis, LTFU <1%
High risk of bias for subjective outcomes (QoL, NYHA, & to lesser extent 6MWD)
Low risk of selection bias as above
But high risk of performance & detection bias due to subjective nature of these outcomes & extra healthcare visits with in-person dietician visits at months 3 & 9 (possibility to identify & mitigate worsening HF or optimize pharmacotherapy)
Trial stopped early due to operational feasibility issues resulting from the COVID-19 pandemic
Other considerations
Generalizability: Limited to individuals already fairly restrictive in dietary sodium
Patients/comparator:
All patients were restricting sodium intake at baseline (~2.2 g/d) compared to the general population (average 4 g/day worldwide). The value of this restriction & ideal targets (e.g. 2-2.5 g/d vs 3-4 g/d) remains unknown.
Notably, these results do not apply to patients who are acutely hospitalized with HF, require escalating diuretic doses, or those with advanced chronic kidney disease
Intervention: Highly transportable/scalable due to use of meal plans & menus rather than prepared meals
Proportion of patients on diuretics used, doses used, & potential changes during the trial have not (yet?) been reported
These results could shed light on the secondary outcomes (e.g. could sodium restriction allow for lower diuretic doses -> less urinary frequency -> improve QoL?)
Similarly, information on changes to HF pharmacotherapy during the trial could offer further insights into these results
Context
Sodium restriction has historically been a cornerstone of HF management for decades due to the role of sodium in contributing to sodium retention and congestive symptoms of HF
However, evidence for sodium retention (& specific targets) has been limited to observational studies and small, inconclusive randomized controlled trials.
The latest Canadian HF guideline recommendations on dietary sodium intake (2017) acknowledged this uncertainty and recommended personalizing daily targets while still recommending 2000-3000 mg of sodium per day (consistent with the usual care group in SODIUM-HF)